Which of the Following Statements About Polar Molecules Are True
Polar molecules are categorized as hydrophobic molecules. All polar molecules have dipole moments.

Solved 1 Point Part 1 Which Of The Following Statements Chegg Com
All molecules in which the central atom is bonded to identical atoms will be a non- polar molecule.
. A because nonpolar molecules have low surface tension Ps. Identify the true statements regarding nucleic acids. III Instantaneous dipoles are most prominent in gases that are non-polar.
The bond dipoles in a polar molecule will cancel. Non-polar molecules can experience dipole-dipole forces. Both statements are true.
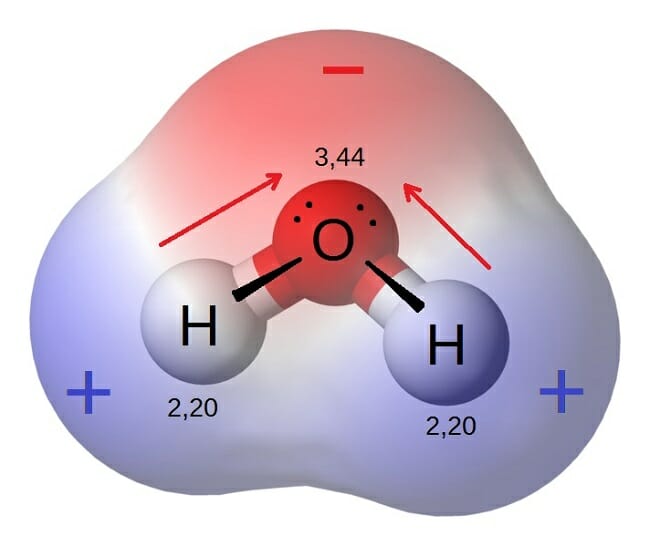
Which of the following statements could be true regarding polar molecules. In order to have a polar molecule there must be unequal distribution of the negatively charged electrons in the orbitals of the molecule. O a A polar molecule is neutral in charge.
A polar molecule will never contain nonpolar bonds. They are generally uncharged molecules. The enthalpy of sublimation is equal to the negative.
- Get Solutions for your doubts here at BYJUS. Statement 1 is true while statement 2 is false. Ion-dipole forces are weaker than Dispersion Forces.
Atop a mountain water boils at a higher temperature. DNA is an example of a nucleic acid. Was this answer helpful.
For a polar molecule which of the following statements is true. Have low surface tension. Statement 1 is false while statement 2 is true.
What we measure is an ensemble and K as-given is for a single molecule. A polar molecule has an uneven distribution of electron density. I and II only.
Which of the following statements is always true concerning polar molecules. Not all molecules having polar bonds are polar. Base sa nabasa ko letter C.
Hydrocarbons are hydrophobic molecules. The bond dipoles in a polar molecule will cancel. A polar molecule will not contain polar bonds.
Which of the following statements is TRUE about nonpolar molecules a. Which of the following statements is not true for polar molecules. 2Polar molecules cannot pass through the polar lipid bilayer 3Neutral molecules pass the membranes through diffusion 4Active transport is exhibited with the expenditure of ATP.
All polar molecules have oppositely charged regions. O a A polar molecule is neutral in charge. Hope your reading your module naka lagay yan sa isang table General properties of polar and non polar molecules.
Previous Devon a vice president of human resources wants to ensure that his companys performance management process is effective so he. Which of the following statements could be true regarding polar molecules. Previous The half-life of a radioactive substance is 357 years.
Molecules must have a single bond to be cis- trans isomers. Choose one or more. They are capable of forming few or no hydrogen bonds.
A polar molecule has an uneven distribution of electron density. Molecules consists of polar bonds can be nonpolar. All polar molecules have hydrogen bonding.
Which of the following is an example of a polar molecule. CH3CH2CH3 has stronger London dispersion forces than does CH4. London dispersion forces are the only type of intermolecular forces that nonpolar molecules exhibit.
A polar molecule is attracted to other molecules because of electrostatic forces. Which of the following statements could be true regarding polar molecules. Which of the following statements could be true regarding polar molecules.
Next When a firm is operating on the downard-sloping. Molecules with the formulas mathrmCH_3 mathrmCH_2 mathrmOH and mathrmC_3 mathrmH_6 mathrmO_2 could be structural isomers. Hydrogen bonds are intermolecular bonds formed between polar molecules.
A polar molecule may have one or more lone pairs. Which of the following statements could be true regarding polar molecules. Diavinad8 and 54 more users found this answer helpful.
CF4 is expected to have a lower boiling at 1 bar than CBr4. A polar molecule will not contain polar bonds. The following statements which are true include.
O A molecule is always polar if it has at least one polar bond. The dipoles are unequally charged either because of the net electronegativity of the molecule or by the shape of the molecule causing the negative dipole. Have high boiling point.
The given statement is False. To be enantiomers a molecule must have at least three different atoms or groups. A The centre of gravity of electrons and protons coincide.
O All three statements are true concerning polar molecules. Which of the following statements is true. All polar molecules have dipole moments.
The oxygen and hydrogen molecules are non-polar molecules. Which of the following statements is not true for polar molecules. The molecules in SO2 g exhibit dipole-dipole intermolecular interactions.
Another example is boron trifluoride B F 3 molecule with triangular planar geometry. A polar molecule will never contain nonpolar bonds. A polar molecule is attracted to other molecules because of electrostatic forces.
How much of a 100 kg sample of the substance will. Which of the following statements about hydrophobic molecules are true about. Hence while v_mp2 prop T it is NOT true that K 12mv2 for one molecule is directly proportional to v_mp2 which is for a collection of molecules.
Another example is carbon. A molecule with a polar bond is always polar. A polar molecule may have one or more lone pairs.
London forces are the only type of intermolecular force found in non polar. They mix well with H20. Pero sagot ko dyan is A.
All polar molecules have oppositely charged regions. An example is linear carbon dioxide molecule C O 2 The individual C O bonds are polar but the molecule on the whole is non polar as individual bond dipoles cancel each other. Select all that apply a.
O A polar molecule is one in which there is an uneven distribution of charge. Nucleotides are the monomers of nucleic acids. Read the following statements and choose the statement that is true.
Science Chemistry QA Library Which of the following statements is true. Which of the following statements is or are true.

Polar And Non Polar Molecules Tmjh 8th Grade Science
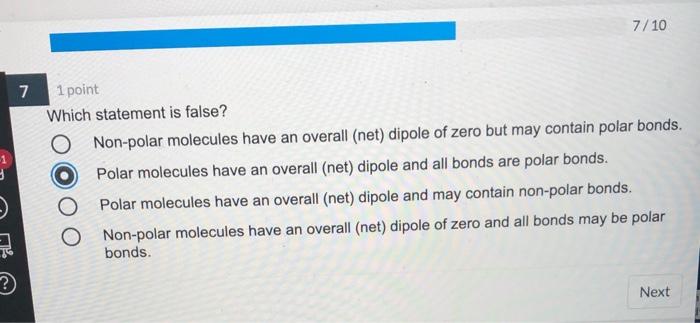
Solved 7 10 7 1 Point Which Statement Is False O Non Polar Chegg Com
0 Response to "Which of the Following Statements About Polar Molecules Are True"
Post a Comment